Ellume Covid-19 Home Test

CNN More than 2 million of Ellumes at-home Covid-19 tests have been recalled by the company due to.
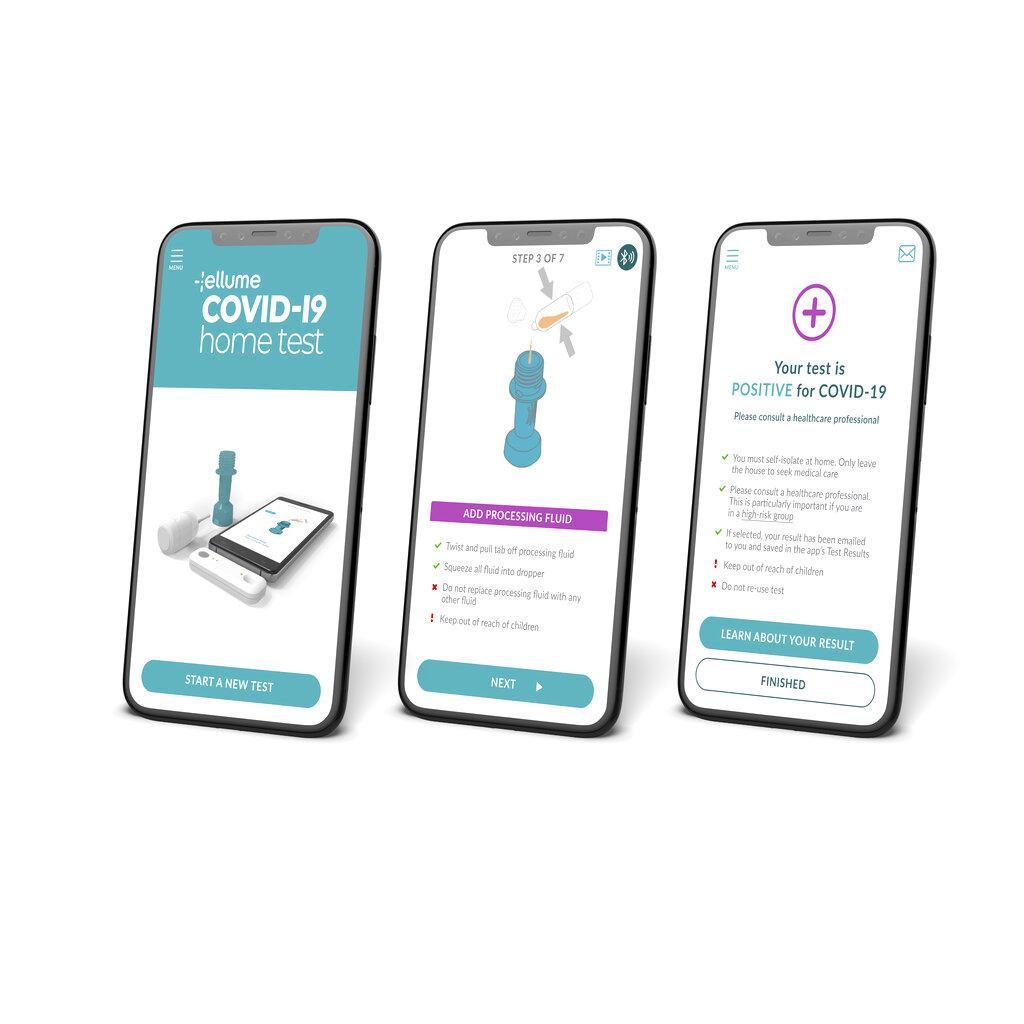
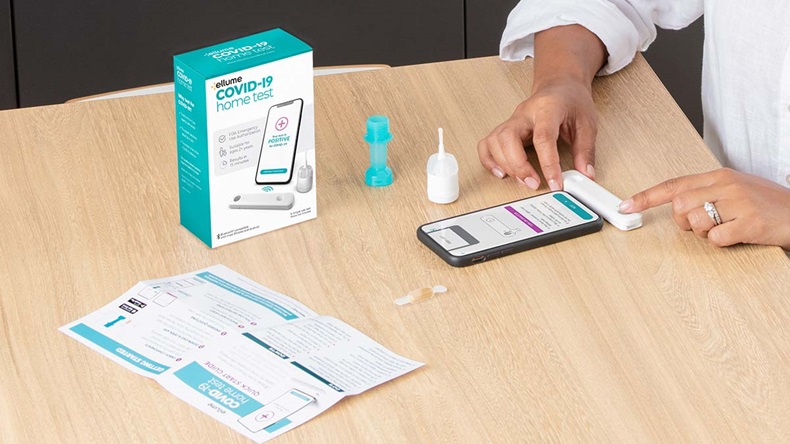
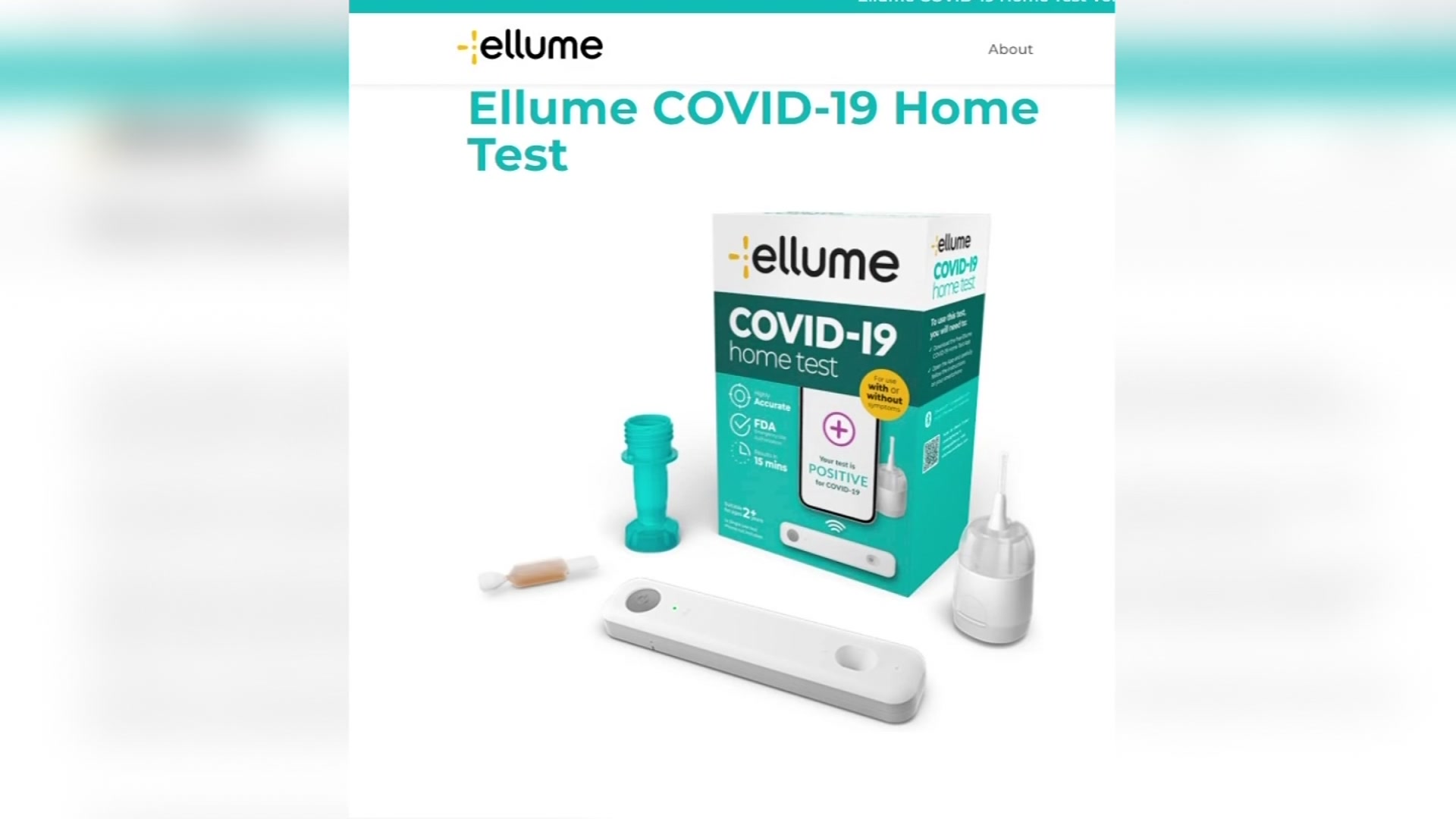
Ellume covid-19 home test. Ellume COVID-19 Home Test with Azova. Ellumes COVID-19 Home Test. Food and Drug Administration has issued a recall of the Ellume COVID-19 Home Test after determining that about 2 million of the tests have the potential for false positive results.
The Cleveland County Health Department will no longer issue the at-home COVID-19 test kit Ellume as it has been recalled by the manufacturer. The Ellume COVID-19 Home Test is an antigen test that works by detecting proteins from the SARS-CoV-2 virus which causes COVID-19. Food and Drug Administration FDA confirmed this issue.
Ellume is encouraging those who have one of. The three testing options now available at CVS include. 1 the federal government announced a 2318 million deal to.
That can be operated entirely by the user at. The Ellume COVID-19 Home Test is a rapid lateral flow immunoassay intended for the qualitative detection of SARS-CoV-2 nucleocapsid antigens from mid-turbinate. In a safety communication on October 5 2021 the US.
Some tests have been removed for safety reasons while others have been removed if the company did not complete an Emergency Use Application request within a reasonable amount of time. Ellume has recalled certain lots of its COVID-19 Home Test after confirming that they may provide false-positive results. Ellume COVID Test Rapid Antigen Home Test Kit Single Use Nasal Swab for COVID-19 1 Pack.
Clinical study the Ellume test showed 96 accuracy for symptomatic individuals. 1 request that the Food and Drug Administration FDA issue. This is just one of several recalls made by the company with the most recent being Oct.